Abstract
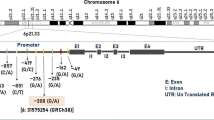
Migraine is a multifactorial disease with various factors, such as genetic polymorphisms and personality traits, but the contribution of those factors is not clear. To clarify the pathogenesis of migraine, the contributions of genetic polymorphisms and personality traits were simultaneously investigated using multivariate analysis. Ninety-one migraine patients and 119 non-headache healthy volunteers were enrolled. The 12 gene polymorphisms analysis and NEO-FFI personality test were performed. At first, the univariate analysis was performed to extract the contributing factors to pathogenesis of migraine. We then extracted the factors that independently contributed to the pathogenesis of migraine using multivariate stepwise logistic regression analysis. Using the multivariate analysis, three gene polymorphisms including monoamine oxidase A (MAOA) T941G, methylenetetrahydrofolate reductase (MTHFR) C677T, and tumor necrosis factor beta (TNF-β) G252Α, and the neuroticism and conscientiousness scores in NEO-FFI were selected as significant factors that independently contributed to the pathogenesis of migraine. Their odds ratios were 1.099 (per point of neuroticism score), 1.080 (per point of conscientiousness score), 2.272 (T and T/T or T/G vs G and G/G genotype of MAOA), 1.939 (C/T or T/T vs C/C genotype of MTHFR), and 2.748 (G/A or A/A vs G/G genotype of TNF-β), respectively. We suggested that multiple factors, such as gene polymorphisms and personality traits, contribute to the pathogenesis of migraine. The contribution of polymorphisms, such as MAOA T941G, MTHFR C677T, and TNF-β G252A, were more important than personality traits in the pathogenesis of migraine, a multifactorial disorder.

Similar content being viewed by others
References
Sakai F, Igarashi H (1997) Prevalence of migraine in Japan: a nationwide survey. Cephalalgia 17:15–22
Mohammad-Zadeh LF, Moses L, Gwaltney-Brant SM (2008) Serotonin: a review. J Vet Pharmacol Ther 31:187–199
Ishii M, Kobayashi S, Ohkura M et al (2009) Inhibitory effect of lomerizine, a prophylactic drug for migraines, on serotonin-induced contraction of the basilar artery. J Pharmacol Sci 111:221–225
Ferrari MD, Odink J, Tapparelli C et al (1989) Serotonin metabolism in migraine. Neurology 39:1239–1242
Nagata E, Shibata M, Hamada J et al (2006) Plasma 5-hydroxytryptamine (5-HT) in migraine during an attack-free period. Headache 46:592–596
Kimball RW, Friedman AP, Vallejo E (1960) Effect of serotonin in migraine patients. Neurology 10:107–111
Lance JW, Anthony M, Hinterberger H (1967) The control of cranial arteries by humoral mechanisms and its relation to the migraine syndrome. Headache 7:93–102
Nyholt DR, Curtain RP, Gaffney PT et al (1996) Migraine association and linkage analyses of the human 5-hydroxytryptamine (5-HT2A) receptor gene. Cephalalgia 16:463–467
Erdal ME, Herken H, Yilmaz M et al (2001) Association of the T102C polymorphism of 5-HT2A receptor gene with aura in migraine. J Neurol Sci 188:99–101
Yilmaz M, Erdal ME, Herken H et al (2001) Significance of serotonin transporter gene polymorphism in migraine. J Neurol Sci 186:27–30
Juhasz G, Zsombok T, Laszik A et al (2003) Association analysis of 5-HTTLPR variants, 5-HT2A receptor gene 102T/C polymorphism and migraine. J Neurogenet 17:231–240
Borroni B, Brambilla C, Liberini P et al (2005) Functional serotonin 5-HTTLPR polymorphism is a risk factor for migraine with aura. J Headache Pain 6:182–184
Marziniak M, Mössner R, Schmitt A et al (2005) A functional serotonin transporter gene polymorphism is associated with migraine with aura. Neurology 64:157–159
Park JW, Han SR, Yang DW et al (2006) Serotonin transporter protein polymorphism and harm avoidance personality in migraine without aura. Headache 46:991–996
Gonda X, Rihmer Z, Juhasz G et al (2007) High anxiety and migraine are associated with the s allele of the 5HTTLPR gene polymorphism. Psychiatry Res 149:261–266
Kowa H, Fusayasu E, Ijiri T et al (2005) Association of the insertion/deletion polymorphism of the angiotensin I-converting enzyme gene in patients of migraine with aura. Neurosci Lett 374:129–131
Kowa H, Yasui K, Takeshima T et al (2000) The homozygous C677T mutation in the methylenetetrahydrofolate reductase gene is a genetic risk factor for migraine. Am J Med Genet 96:762–764
Schurks M, Zee RY, Buring JE et al (2010) MTHFR 677C– > T and ACE D/I polymorphisms and migraine attack frequency in women. Cephalalgia 30:447–456
Colson NJ, Lea RA, Quinlan S et al (2004) The estrogen receptor 1 G594A polymorphism is associated with migraine susceptibility in two independent case/control groups. Neurogenetics 5:129–133
Oterino A, Pascual J, Ruiz de Alegría C et al (2006) Association of migraine and ESR1 G325C polymorphism. Neuroreport 17:61–64
Asuni C, Stochino ME, Cherchi A et al (2009) Migraine and tumour necrosis factor gene polymorphism. An association study in a Sardinian sample. J Neurol 256:194–197
Breslau N, Andreski P (1995) Migraine, personality, and psychiatric comorbidity. Headache 35:382–386
Moschiano F, D’Amico D, Canavero I et al (2011) Migraine and depression: common pathogenetic and therapeutic ground? Neurol Sci 32(suppl 1):S85–S88
Headache Classification Subcommittee of the International Headache Society (2004) The international classification of headache disorders: 2nd edition. Cephalalgia 24(suppl 1):9–160
Aiba S, Tatsumoto M, Saisu A et al (2010) Prevalence of typical migraine aura without headache in Japanese ophthalmology clinics. Cephalalgia 30:962–967
Costa PT Jr, McCrae RR (1992) Revised NEO personality inventory (NEO-PI-R) and NEO five-factor inventory (NEO-FFI) professional manual. Psychological Assessment Resources, Odessa
Murakami F, Shimomura T, Kotani K et al (1999) Anxiety traits associated with a polymorphism in the serotonin transporter gene regulatory region in the Japanese. J Hum Genet 44:15–17
Kaiser R, Muller-Oerlinghausen B, Filler D et al (2002) Correlation between serotonin uptake in human blood platelets with the 44-bp polymorphism and the 17-bp variable number of tandem repeat of the serotonin transporter. Am J Med Genet 114:323–328
Joober R, Benkelfat C, Brisebois K et al (1999) T102 polymorphism in the 5-HT2A gene and schizophrenia: relation to phenotype and drug response variability. J Psychiatry Neurosci 24:141–146
MaassenVanDenBrink A, Vergouwe MN, Ophoff RA et al (1998) 5-HT1B receptor polymorphism and clinical response to sumatriptan. Headache 38:288–291
Deckert J, Catalano M, Syagailo YV et al (1999) Excess of high activity monoamine oxidase a gene promoter alleles in female patients with panic disorder. Hum Mol Genet 8:621–624
Guo G, Ou XM, Roettger M et al (2008) The VNTR 2 repeat in MAOA and delinquent behavior in adolescence and young adulthood: associations and MAOA promoter activity. Eur J Hum Genet 16:626–634
Hotamisligil GS, Breakefield XO (1991) Human monoamine oxidase a gene determines levels of enzyme activity. Am J Hum Genet 49:383–392
Frosst P, Blom HJ, Milos R et al (1995) A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase. Nat Genet 10:111–113
Rigat B, Hubert C, Corvol P et al (1992) PCR detection of the insertion/deletion polymorphism of the human angiotensin converting enzyme gene (DCP1) (dipeptidyl carboxypeptidase 1). Nucleic Acids Res 20:1433
Colson NJ, Lea RA, Quinlan S et al (2006) No role for estrogen receptor 1 gene intron 1 Pvu II and exon 4 C325G polymorphisms in migraine susceptibility. BMC Med Genet 7:12
Curran JE, Lea RA, Rutherford S et al (2001) Association of estrogen receptor and glucocorticoid receptor gene polymorphisms with sporadic breast cancer. Int J Cancer 95:271–275
Sarkar G, Kapelner S, Grandy DK et al (1991) Direct sequencing of the dopamine D2 receptor (DRD2) in schizophrenic’s reveals three polymorphisms but no structural change in the receptor. Genomics 11:8–14
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Erlbaum Associates, Hillsdale
Kimura K, Iguchi Y, Shibazaki K et al (2008) Hemorrhagic transformation of ischemic brain tissue after t-PA thrombolysis as detected by MRI may be asymptomatic, but impair neurological recovery. J Neurol Sci 272:136–142
Lee TC, Wang HP, Chiu HM et al (2010) Male gender and renal dysfunction are predictors of adverse outcome in nonpostoperative ischemic colitis patients. J Clin Gastroenterol 44:e96–e100
Ishii M, Sakairi Y, Hara H et al. (2011) Negative predictors of clinical response to triptans in patients with migraine. Neurol Sci. doi:10.1007/s10072-011-0716-z
Mattsson P, Ekselius L (2002) Migraine, major depression, panic disorder, and personality traits in women aged 40–74 years: a population-based study. Cephalalgia 22:543–551
Mongini F, Fassino S, Rota E et al (2005) The temperament and character inventory in women with migraine. J Headache Pain 6:247–249
Mikolajczyk E, Zietek J, Samochowiec A et al (2008) Personality dimensions measured using the temperament and character inventory (TCI) and NEO-FFI on a Polish sample. Int J Methods Psychiatr Res 17:210–219
Ozelius L, Hsu YP, Bruns G et al (1988) Human monoamine oxidase gene (MAOA): chromosome position (Xp21–p11) and DNA polymorphism. Genomics 3:53–58
Hinds HL, Hendriks RW, Craig IW et al (1992) Characterization of a highly polymorphic region near the first exon of the human MAOA gene containing a GT dinucleotide and a novel VNTR motif. Genomics 13:896–897
Fan M, Liu B, Jiang T et al (2010) Meta-analysis of the association between the monoamine oxidase-A gene and mood disorders. Psychiatr Genet 20:1–7
Taylor LM Jr, DeFrang RD, Harris EJ Jr et al (1991) The association of elevated plasma homocyst(e)ine with progression of symptomatic peripheral arterial disease. J Vasc Surg 13:128–136
Prasad K (1999) Homocysteine, a risk factor for cardiovascular disease. Int J Angiol 8:76–86
Blount BC, Mack MM, Wehr CM et al (1997) Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implications for cancer and neuronal damage. Proc Natl Acad Sci USA 94:3290–3295
Duthie SJ, Narayanan S, Brand GM et al (2002) Impact of folate deficiency on DNA stability. J Nutr 132(suppl):2444S–2449S
de Bree A, Verschuren WM, Bjorke-Monsen AL et al (2003) Effect of the methylenetetrahydrofolate reductase 677C– > T mutation on the relations among folate intake and plasma folate and homocysteine concentrations in a general population sample. Am J Clin Nutr 77:687–693
Moschiano F, D’Amico D, Usai S et al (2008) Homocysteine plasma levels in patients with migraine with aura. Neurol Sci 29(suppl 1):S173–S175
Rubino E, Ferrero M, Rainero I et al (2009) Association of the C677T polymorphism in the MTHFR gene with migraine: a meta-analysis. Cephalalgia 29:818–825
Todt U, Freudenberg J, Goebel I et al (2006) MTHFR C677T polymorphism and migraine with aura. Ann Neurol 60:621–622
Kaunisto MA, Kallela M, Hamalainen E et al (2006) Testing of variants of the MTHFR and ESR1 genes in 1,798 Finnish individuals fails to confirm the association with migraine with aura. Cephalalgia 26:1462–1472
Oterino A, Toriello M, Valle N et al (2010) The relationship between homocysteine and genes of folate-related enzymes in migraine patients. Headache 50:99–168
Gilbody S, Lewis S, Lightfoot T (2007) Methylenetetrahydrofolate reductase (MTHFR) genetic polymorphisms and psychiatric disorders: a HuGE review. Am J Epidemiol 165:1–13
Miller AL (2008) The methylation, neurotransmitter, and antioxidant connections between folate and depression. Altern Med Rev 13:216–226
Koslow SH, Butler IJ (1977) Biogenic amine synthesis defect in dihydropteridine reductase deficiency. Science 198:522–523
Miwa S, Watanabe Y, Hayashi O (1985) 6R-L-erythro-5, 6, 7, 8-tetrahydrobiopterin as a regulator of dopamine and serotonin biosynthesis in the rat brain. Arch Biochem Biophys 239:234–241
Trabace S, Brioli G, Lulli P et al (2002) Tumor necrosis fator gene polymorphism in migraine. Headache 42:341–345
Ghosh J, Joshi G, Pradhan S et al (2010) Investigation of TNFA 308G > A and TNFB 252G > A polymorphisms in genetic susceptibility to migraine. J Neurol 257:898–904
Abraham LJ, French MA, Dawkins RL (1993) Polymorphic MHC ancestral haplotypes affect the activity of tumour necrosis factor-alpha. Clin Exp Immunol 92:14–18
Grimble RF, Howell WM, O’Reilly G et al (2002) The ability of fish oil to suppress tumor necrosis factor α production by peripheral blood mononuclear cells in healthy men is associated with polymorphisms in genes that influence tumor necrosis factor α production. Am J Clin Nutr 76:454–459
Messer G, Spengler U, Jung MC et al (1991) Polymorphic structure of the tumor necrosis factor (TNF) locus: an NcoI polymorphism in the first intron of the human TNF-β gene correlates with a variant amino acid in position 26 and a reduced level of TNF-β production. J Exp Med 173:209–219
Perini F, D’Andrea G, Galloni E et al (2005) Plasma cytokine levels in migraineurs and controls. Headache 45:926–931
Ophoff RA, Terwindt GM, Vergouwe MN et al (1996) Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell 87:543–552
De Fusco M, Marconi R, Silvestri L et al (2003) Haploinsufficiency of ATP1A2 encoding the Na+/K+ pump α2 subunit associated with familial hemiplegic migraine type 2. Nat Genet 33:192–196
Acknowledgments
This study was supported in part by a grant of Private University High Technology Research Center Project matching fund subsidy from Ministry of Education, Culture, Sport, Science, and Technology, Japan (MEXT).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishii, M., Shimizu, S., Sakairi, Y. et al. MAOA, MTHFR, and TNF-β genes polymorphisms and personality traits in the pathogenesis of migraine. Mol Cell Biochem 363, 357–366 (2012). https://doi.org/10.1007/s11010-011-1188-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-011-1188-4